|
The f-block is a fundamental part of the periodic table, yet it is usually omitted from A-level specifications. What are the f-block elements and what makes them so unique? Read on to find out more... What is the f-block? The f-block is a group of 28 elements, conventionally situated below the periodic table. However this position is misleading; the elements are part of the main table, but to make the periodic table fit on a page (and be readable), they are often removed and placed on the bottom. 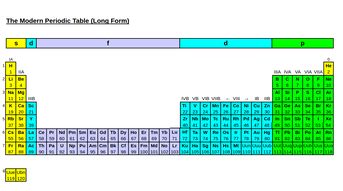 The 'long' form of the periodic table shows where the elements are meant to be - right in the middle. Each row is 14 elements long, with the top row being called the lanthanides (named after the first element in the row, lanthanum) and the bottom row being called the actinides (named after the first element in the row, actinium). The elements in these rows have an f-based valence shell which can contain up to 14 electrons, or 7 orbitals. When writing the electron configuration of these elements, the Aufbau principle must be used: 4f fills after 6s and 5f fills after 7s. For example, Neodymium (Nd, atomic number 60) has the electron configuration: 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 4f4 6s2 The LanthanidesProperties: One of the most remarkable properties of the lanthanides is that, despite being in the same row, they nearly always have the same oxidation state of +3. This feature is due to the stability provided by this oxidation state, and so the 4th ionisation energies of lanthanides tend to be extremely high. Another feature worth mentioning is the concept of lanthanide contraction. This is the greater than expected decrease in ionic and atomic radii as you move across the period. This occurs due to a large increase in nuclear charge yet very little additional electron shielding, which allows the nucleus to exert a greater 'pull' on the outer electrons. This concept feature can also be found in the actinides. The basicity (ease at which an atom will lose electrons) decreases across the period. The lanthanides also have magnetic properties, as the only diamagnetic (having no unpaired electrons, so not magnetic) ions are La3+, Lu3+, Yb2+ and Ce4+. The rest are all paramagnetic (having unpaired electrons and hence magnetic properties).  Uses: The relative stability of the +3 oxidation state mentioned above means that ions such as Eu2+ and Sm2+ are thermodynamically unstable compared with the +3 ions. Therefore, these ions can be used in reactions as strong reducing agents (loses an electron to reduce another element), especially in reactions where organic radicals are needed. Like the actindies, they can also be used in nuclear power. Their oxides can act as dilutents (diluting agents) in nuclear fields, whilst the hydrides can be hydrogen-moderator characters. Some of the elements, such as Tm, can be used as a portable source of x-rays, and others are sources of radiation, such as Eu. The ActinidesProperties: All of the actinides exhibit radioactivity, due to their large size (lead-208 is the heaviest known element to be stable). Whilst some isotopes are more radioactive than others, this does lead to many uses in nuclear powerplants and other items, such as smoke detectors. Unlike the Lanthanides, there is no set oxidation state for the actinides, with the usual variance being between +2 and +7. This is due to the 5f, 6d and 7s subshells being very close together, making it easy to remove electrons from more than one. Many of the actinides have been made synthetically, and are short-lived when made. In nature, they often occur as sea water or minerals. All have a silver sheen in metallic form.  Uses: Plutonium and Uranium are used as both fuels for nuclear power plants, and also nuclear bombs. The 238- Plutonium isotope also was in one of the generators on the Apollo-12 Lunar mission, powers pacemakers for the heart and was a power source for orbiting satellites taking photos of Jupiter The 241- Americium isotope also powers smoke detectors by emitting alpha radiation that is picked up by electrodes. Smoke stops the alpha particles from reaching the electrodes, triggering an alarm. Comments are closed.
|
ChemIzzyA chemistry-mad sixth form student based in the UK, helping you with tips, revision and further reading. Categories
All
|
